
HypericinCAS No.:548-04-9
|
||||||||||
 |
|
|
||||||||

| Catalogue No.: | BP0752 |
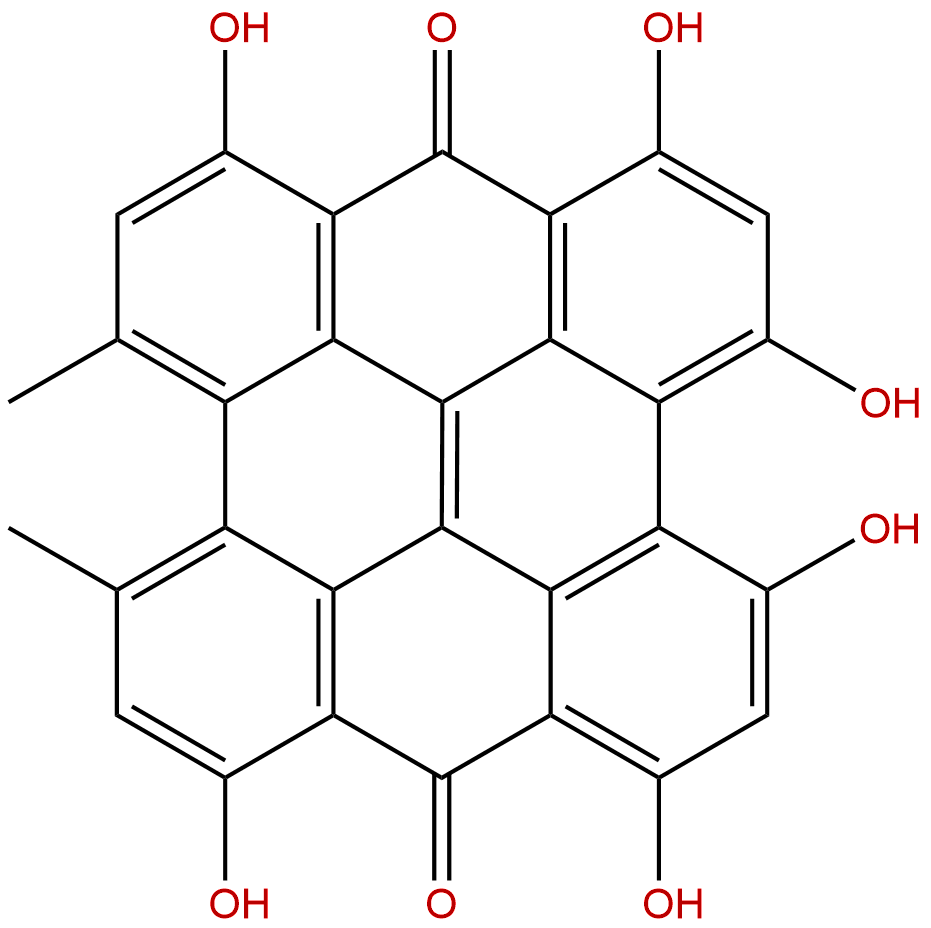
| Formula: | C30H16O8 |
| Mol Weight: | 504.45 |
Synonym name: Mycoporphyrin
Catalogue No.: BP0752
Cas No.: 548-04-9
Formula: C30H16O8
Mol Weight: 504.45
Botanical Source: the mealy bug Nipaecoccus aurilanatus. Widespread in Hypericum spp. esp. Hypericum perforatum (St John's wort)
Purity: 95%~99%
Analysis Method: HPLC-DAD
Identification Method: Mass, NMR
Can be supplied from milligrams to grams, up to kilograms.
Inquire for bulk scale.
It can be produced in GMP factory as per customer's requiring.
About Hypericn
Solubility: good soluble in ethanol, methanol, pyridine, aceton, ethyl acetate, butanone, DMSO and aqueous alkali solutions; and insoluble in water and methylene chloride.
Hypericin, a photochemical extracted from St. Johns Wort (Hypericum perforatum) and related species. Hypericin has been shown to have potent, broad spectrum antimicrobial activity. Hypericin is an aromatic polycyclic anthrone, a class of colored or pigmented chemical substances which have photosensitizing activity. In both in vitro (laboratory) and in vivo (animal) studies, low, non-toxic doses of hypericin significantly inhibited the replication of several viruses, including HIV, influenza A, cytomegalovirus (CMV), Herpes simplex 1 and 2 (HSV-1 and HSV-2), and Epstein-Barr virus (EBV). Hypericin and its chemical relative, pseudohypericin, produce antiviral activity through a different mechanism of action than do AZT and other nucleoside antiviral agents. Hypericin does not appear to directly alter the activity of reverse transcriptase although it does block the formation of HIV synctium. Recent findings have shown that the antiretroviral action of this compound disrupts uncoating of the lipid envelope of both DNA and RNA viruses, thus preventing infected cells from releasing HIV copies. Theoretically, hypericin and AZT, in combination, may have synergistic antiviral effects against HIV. On the other hand, hypericin actually may increase the toxicity of antiretroviral nucleosides such as AZT, ddI, or ddC.
Traditionally, extracts of St. Johns Wort (which contain hypericin) have been used as an antidepressant, possibly by acting as a MAO inhibitor. The psychotropic effects attributed to hypericin in St. Johns Wort extract suggest that the pigment compound can cross the blood brain barrier (possibly treating neuropsychological symptoms such as dementia). Laboratory investigations indicate that hypericin may be beneficial as an HIV therapy. However, its administration should be carefully monitored by a physician. The levels of hypericin found in most commercially available extracts of St. Johns Wort generally are not sufficient to be therapeutically effective against viral infections.
Liver function should be tested periodically in persons taking hypericin. Also, extreme photosensitivity has been observed in a few cases of people taking this high doses (in excess of 10 mg per day) of this compound. Finally, there is a very small possibility that adverse reactions could occur on occasion between hypericin and other foods or drugs which interfere with MAO inhibitors.
Dosage
Clinical trials currently being conducted by Dr. Bihari in New York City recommend the following regimen. For the first two weeks, patients take one 10 mg. capsule once a day for two weeks. During the second two weeks, the dosage alternates between one 10 mg. capsule per day on the first day, two 10 mg. capsules on the second day, one 10 mg. capsule on the third day, and so forth. The ultimate dosage is given during the fifth and sixth weeks when patients are given two 10 mg. capsules per day.
Future regimens for optimal dosage are presently being devised by researchers and clinicians involved in pilot trials of hypericin for the treatment of HIV. It has been proposed that beta-carotene, a known free radical quencher, be administered in combination with hypericin. Theoretically, this might reduce any toxic side-effects associated with hypericin’s generation of free radicals. Some investigators, however, have warned that quenchers also may lessen the anti-viral properties of hypericin. The role of beta-carotene and other free-radical scavengers in hypericin therapy deserves further clarification.
HPLC of Hypericin
