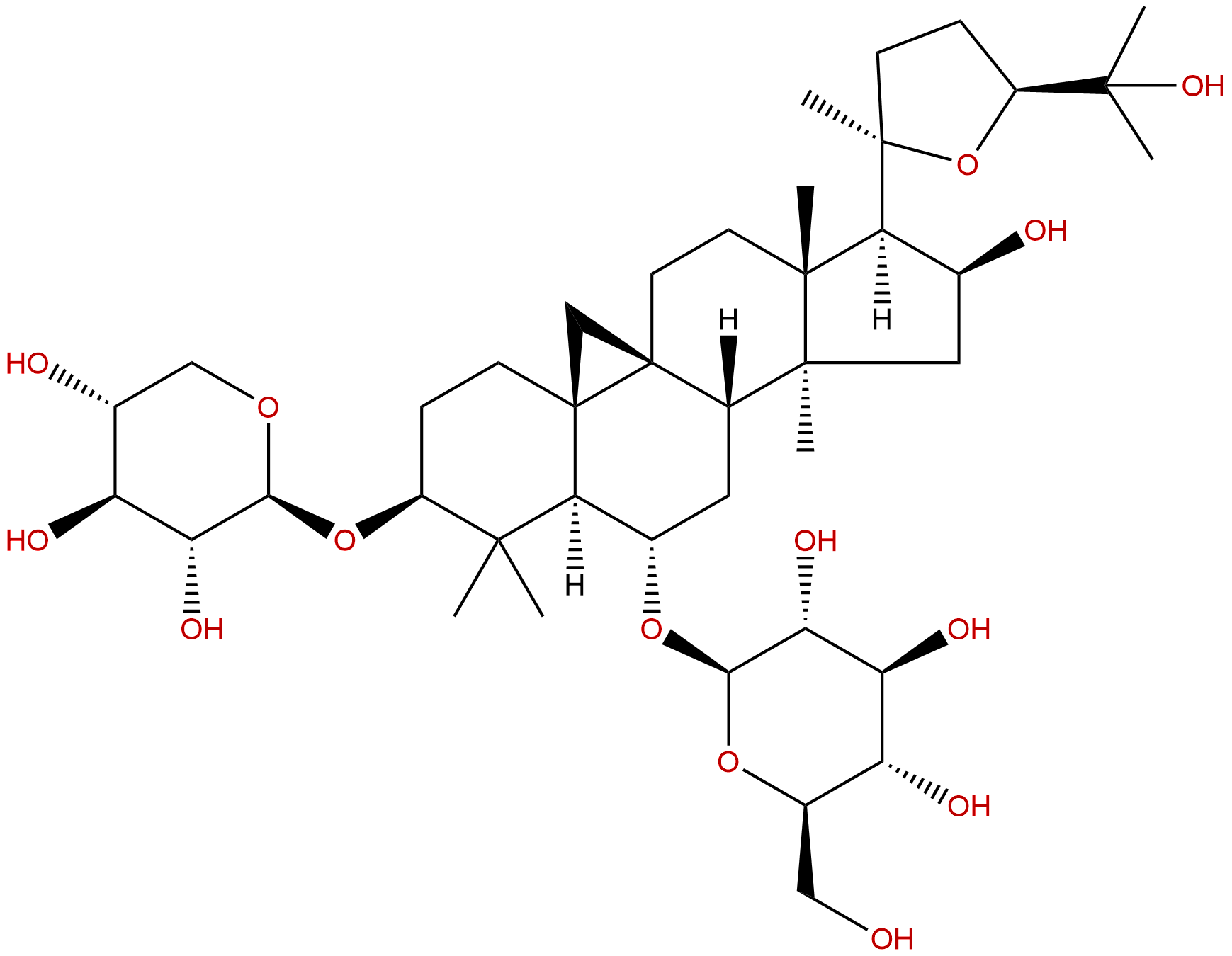
Astragaloside IVCAS No.:84687-43-4
|
||||||||||
 |
|
|
||||||||

| Catalogue No.: | BP0213 |
| Formula: | C41H68O14 |
| Mol Weight: | 784.981 |
Product name: Astragaloside IV
Synonym name: Astrasieversianin XIV; Cyclosieversioside F; Cyclosiversioside F; Astramembrannin I; Astragaloside A
Catalogue No.: BP0213
Cas No.: 84687-43-4
Formula: C41H68O14
Mol Weight: 784.981
Botanical Source: Astragalus membranaceus, Astragalus sieversianus and Astragalus pterocephalus
Physical Description: White powder
Type of Compound: Triterpenoids
Purity: 95%~99%
Analysis Method: HPLC-DAD or/and HPLC-ELSD
Identification Method: Mass, NMR
Packing: Brown vial or HDPE plastic bottle
Storage: Store in a well closed container, protected from air and light. Put into refrigerate or freeze for long term storage.
Whenever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20℃. Generally, these will be useable for up to two weeks.
The product could be supplied from milligrams to grams, up to kilograms
Inquire for bulk scale.
Descriptions:
Astragaloside IV, a saponin isolated from Astragalus membranaceus, has been shown to protect the myocardium against ischemia/reperfusion injury, it also has beneficial effect in H/R-induced injury may be related to normalization of SR Ca2+ pump expression and, thus, it may prevent the depression in SR Ca2+ handling.[1]
Astragaloside IV synergizes with ferulic acid to inhibit renal tubulointerstitial fibrosis in rats with obstructive nephropathy.[2]
Astragaloside IV attenuates glycated albumin-induced epithelial-to-mesenchymal transition by inhibiting oxidative stress in renal proximal tubular cells.[3]
Astragaloside IV can reduce phosphorylation of JNK and ERK1/2 induced by complement membranous attack complex, the mechanism of Astragalus membranaceus in the treatment of membranous nephropathy (MN) may be related to its attenuation of podocyte injury through regulation of cytoskeleton and mitogen activated protein kinase. [4]
Astragaloside IV can reduce blood pressure and triglyceride levels in fructose-fed rats and high dose of astragaloside IV also improves glucose tolerance and endothelium-dependent vasorelaxation, the mechanism is associated with increased levels of aortic NOx and cGMP and is abrogated by blockade of nitric oxide synthase with NG-nitro-l-arginine methyl ester (l-NAME), suggests that astragaloside IV may be useful in ameliorating food-induced metabolic syndrome.[5]
Astragaloside IV attenuates inflammatory cytokines by inhibiting TLR4/NF-кB signaling pathway in isoproterenol-induced myocardial hypertrophy, and attenuates Toll-like receptor 4 expression via NF-κB pathway under high glucose condition in mesenchymal stem cells.[6,7]
Astragaloside IV can inhibit adenovirus replication and apoptosis in A549 cells in vitro.[8]
Astragaloside IV can inhibit doxorubicin-induced cardiomyocyte apoptosis mediated by mitochondrial apoptotic pathway via activating the PI3K/Akt pathway.[9]
References:
[1] Xu X L, Chen X J, Ji H, et al. Pharmacology, 2008, 81(4):325-32.
[2] Meng L, Tang J, Wang Y, et al. Brit J Pharmacol, 2011, 162(8):1805-18.
[3] Qi W, Niu J, Qin Q, et al. Cell Stress & Chaperon, 2014, 19(1):105-14.
[4] Rong Z, Deng Y, Chen Y, et al. Phytother Res, 2012, 26(6):892-8.
[5] Zhang N, Wang X H, Mao S L. Molecules, 2011, 16(5):3896-907.
[6] Yang J, Wang H X, Zhang Y J, et al. J Ethnopharmacol, 2013, 150(3):1062–70.
[7] Li M, Yu L, She T, et al. Eur J Pharmacol, 2012, 696(1-3):203-9.
[8] Shang L, Qu Z, Sun L, et al. J Pharm Pharmacol, 2011, 63(5):688-94.
[9] Jia Y, Zuo D, Li Z, et al. Chem Pharm Bull, 2014, 62(1):45-53.
[10] Liu Q, Zhongdong L I, Liu J, et al. Chinese Journal of Modern Applied Pharmacy, 2010(04):355-8.
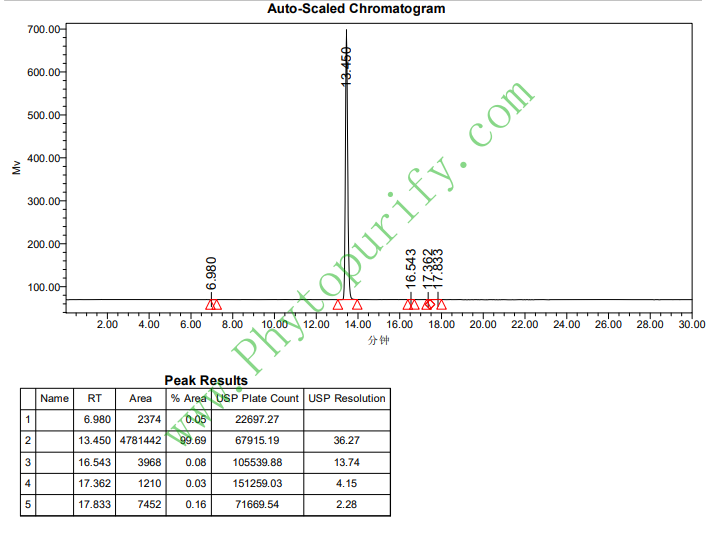
HPLC of Astragaloside IV