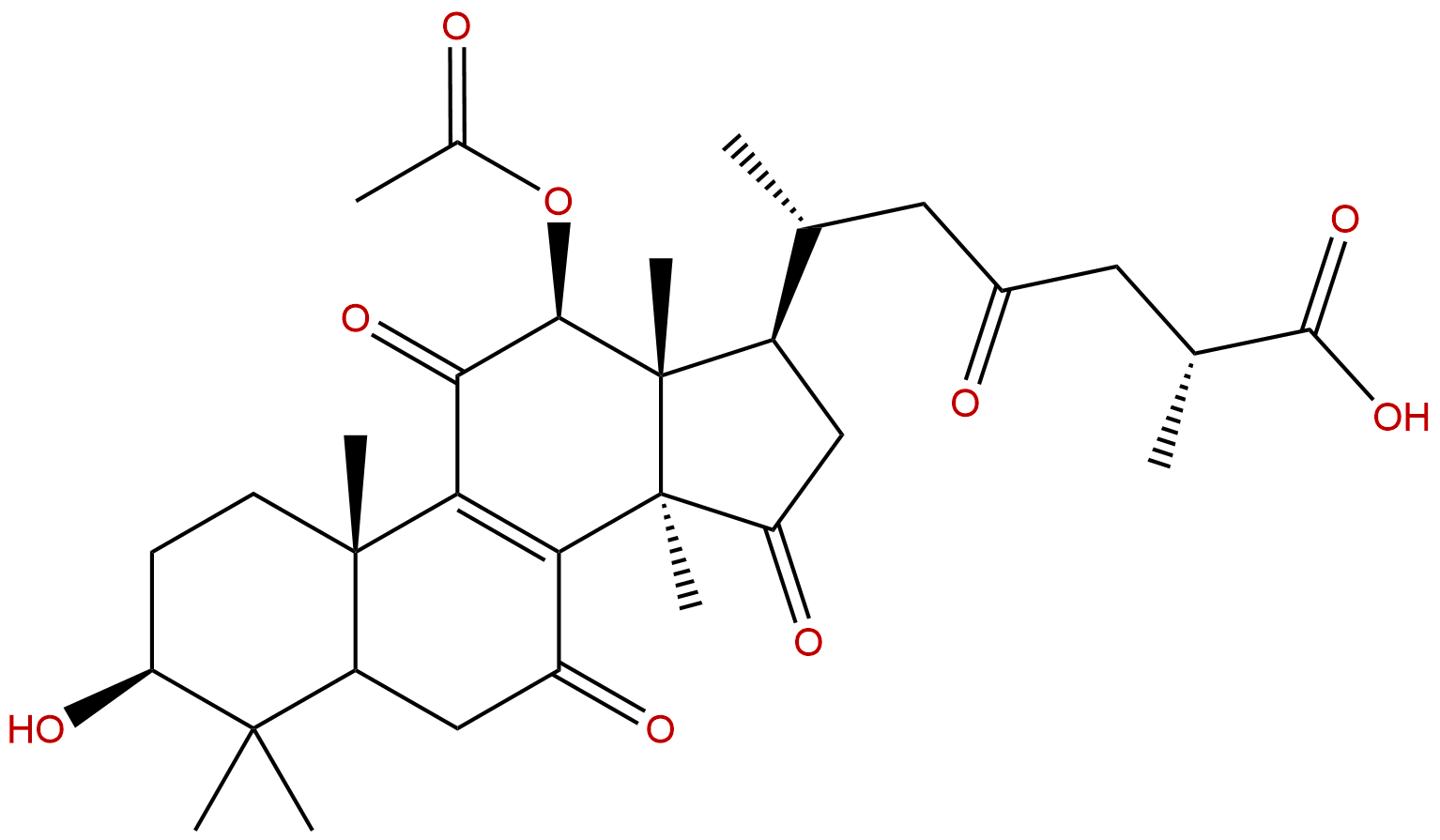
Ganoderic Acid H Descrtption
Product name: Ganoderic Acid H
Synonym name: Ganoderic acid C
Catalogue No.: BP0620
Cas No.: 98665-19-1
Formula: C32H44O9
Mol Weight: 572.695
Botanical Source: From Ganoderma lucidum (reishi)
Physical Description: Powder
Type of Compound: Triterpenoids
Purity: 95%~99%
Analysis Method: HPLC-DAD or/and HPLC-ELSD
Identification Method: Mass, NMR
Packing: Brown vial or HDPE plastic bottle
Storage: Store in a well closed container, protected from air and light. Put into refrigerate or freeze for long term storage.
Whenever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20℃. Generally, these will be useable for up to two weeks.
The product could be supplied from milligrams to grams, up to kilograms
Inquire for bulk scale.
Descriptions:
Ganoderic acid H( GA-H )and ganoderic acid A(GA-A ) suppress growth (cell proliferation and colony formation) and invasive behavior (adhesion, migration and invasion) of MDA-MB-231 cells; they mediate their biological effects through the inhibition of transcription factors AP-1 and NF-kappaB, resulting in the down-regulation of expression of Cdk4 and the suppression of secretion of uPA, respectively; furthermore, the activity of ganoderic acids is linked to the hydroxylation in the position 7 and 15 (GA-A) and 3 (GA-H) in their triterpene lanostane structure; so hydroxylated triterpenes from G. lucidum could be promising natural agents for the therapy of invasive breast cancers.[1]
References:
[1] Jiang J, Grieb B, Thyagarajan A, et al. Int J Mol Med, 2008, 21(5):577-84.
[2] Wang X M, Guan S H, Liu R X, et al. Journal of Shenyang Pharmaceutical University, 2007, 24(07):407-9.