
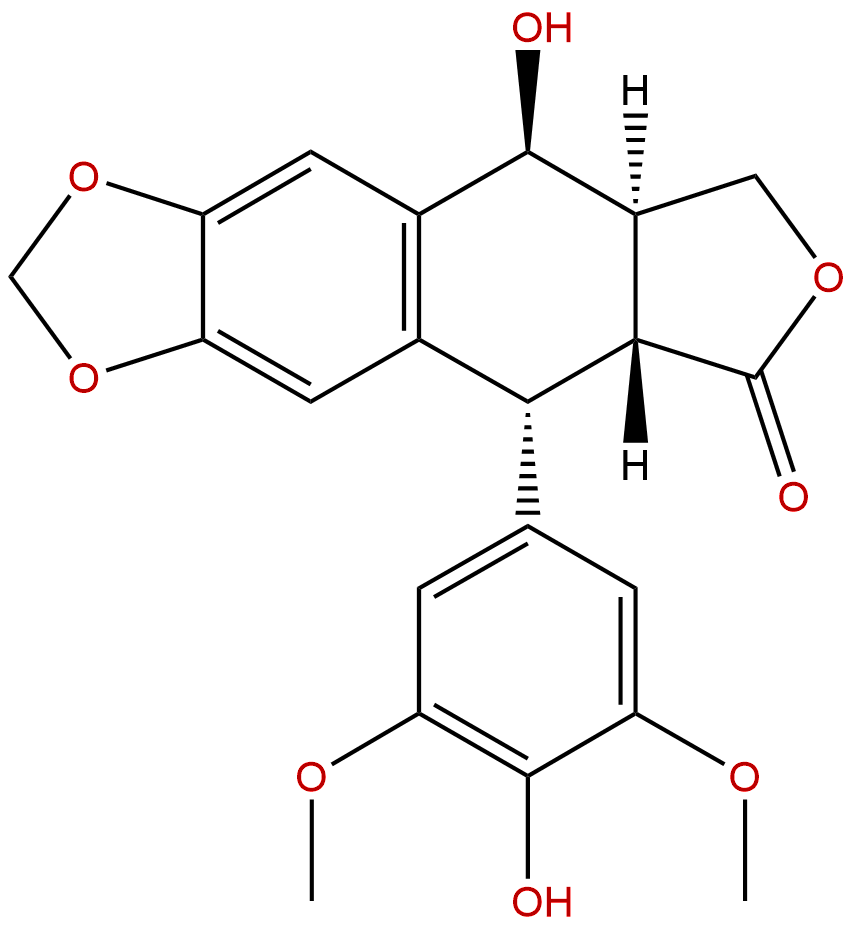
4'-DemethylepipodophyllotoxinCAS No.:6559-91-7
|
||||||||||
 |
|
|
||||||||

| Catalogue No.: | BP0075 |
| Formula: | C21H20O8 |
| Mol Weight: | 400.383 |
Synonym name:
Catalogue No.: BP0075
Cas No.: 6559-91-7
Formula: C21H20O8
Mol Weight: 400.383
Botanical Source:
Purity: 95%~99%
Analysis Method: HPLC-DAD or/and HPLC-ELSD
Identification Method: Mass, NMR
Packing: Brown vial or HDPE plastic bottle
Can be supplied from milligrams to grams.
For Reference Standard and R&D, Not for Human Use Directly.
Inquire for bulk scale.
Description:
4'-Demethylepipodophyllotoxin is an antimitotic agent which binds to monomeric tubulin, preventing micro-tubule polymerization. It is a potent inhibitor of microtubule assembly.
References:
Bioorg Med Chem. 2014 Jun 1;22(11):2998-3007.
A rational design strategy of the novel topoisomerase II inhibitors for the synthesis of the 4-O-(2-pyrazinecarboxylic)-4'-demethylepipodophyllotoxin with antitumor activity by diminishing the relaxation reaction of topoisomerase II-DNA decatenation.
A rational design strategy of the novel podophyllum topoisomerase II (Topo II) inhibitors for the synthesis of the esterification and amidation substituted 4'-Demethylepipodophyllotoxin (DMEP) derivates was developed in order to discover the potential antitumor prodrug. Firstly, according to the structure-activity relationship, drug combination principle and bioisosterism, the -COO- and the -NH- bond substituents at the 4 position of cycloparaffin would be a great modification direction to improve antitumor activity of 4'-Demethylepipodophyllotoxin (DMEP). Secondly, from the prodrug principle view, the esterification and amidation at the C-4 position of DMEP would be two useful structure modifications for improve solubility. Thirdly, from the activity pocket in Topo II-DNA cleavage complex point of view, a series of heterocyclic with pharmacological activity were chosen as module for improving antitumor activity by binding with Topo II. Finally, nine novel esterification and amidation DMEP derivates were designed and synthesized for the potential Topo II inhibitors with the superior biological activity.
METHODS AND RESULTS:
All the novel compounds exhibited promising in vitro antitumor activity, especially 4-O-(2-pyrazinecarboxylic)-4'-Demethylepipodophyllotoxin (compound 1). The antitumor activity of compound 1 against tumor cell line HeLa (i.e., the IC50 value of 0.60 ± 0.20 μM), A549 (i.e., the IC50 value of 3.83 ± 0.08 μM), HepG2 (i.e., the IC50 value of 1.21 ± 0.05 μM), and BGC-823 (i.e., the IC50 value of 4.15 ± 1.13 μM) was significantly improved by 66, 16, 12, and 6 times than that of the clinically important podophyllum anticancer drug etoposide (i.e., the IC50 values of 15.32 ± 0.10, 59.38 ± 0.77, 67.25 ± 7.05, and 30.74 ± 5.13 μM), respectively.
CONCLUSIONS:
Compound 1 could arrest HeLa cell cycle G2/M and induce apoptosis by strongly diminishing the relaxation reaction of Topo II-DNA decatenation. The correctness of rational drug design was strictly demonstrated by the bioactivity test.