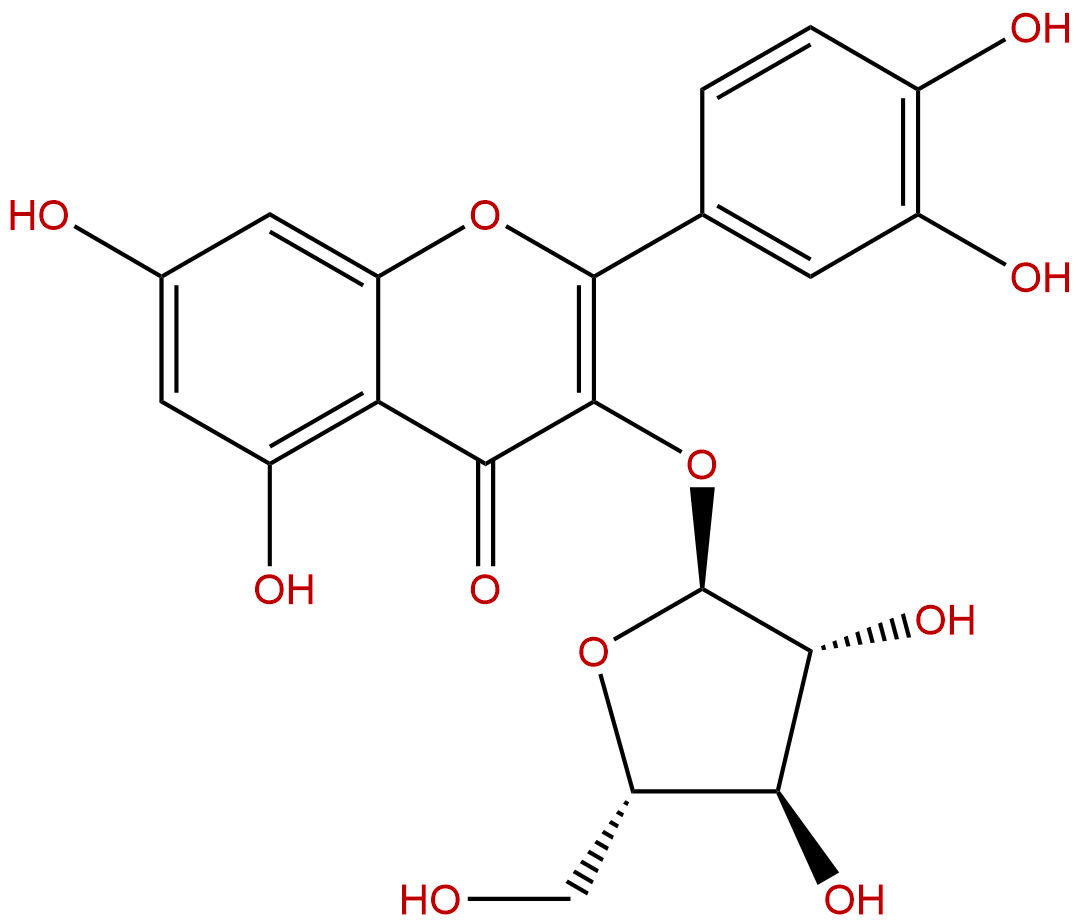
AvicularinCAS No.:572-30-5
|
||||||||||
 |
|
|
||||||||

| Catalogue No.: | BP0223 |
| Formula: | C20H18O11 |
| Mol Weight: | 434.353 |
Synonym name: Avicularoside; Fenicularin
Catalogue No.: BP0223
Cas No.: 572-30-5
Formula: C20H18O11
Mol Weight: 434.353
Botanical Source: Polygonum aviculare, Andromeda polifolia, Vaccinium myrtillus, Juglans regia and other plants
Purity: 95%~99%
Analysis Method: HPLC-DAD or/and HPLC-ELSD
Identification Method: Mass, NMR
Packing: Brown vial or HDPE plastic bottle
Can be supplied from milligrams to grams.
For Reference Standard and R&D, Not for Human Use Directly.
Inquire for bulk scale.
Description:
Avicularin exhibits anti-inflammatory activity through the suppression of ERK signaling pathway in LPS-stimulated RAW 264.7 macrophage cells; it inhibits the accumulation of the intracellular lipids by decreasing C/EBPα-activated GLUT4-mediated glucose uptake in adipocytes and potently inhibiting fatty acid synthase.
References:
Biomol Ther (Seoul). 2012 Nov;20(6):532-7.
Avicularin Inhibits Lipopolysaccharide-Induced Inflammatory Response by Suppressing ERK Phosphorylation in RAW 264.7 Macrophages.
suppresAvicularin, quercetin-3-α-L-arabinofuranoside, has been reported to possess diverse pharmacological properties such as anti-inflammatory and anti-infectious effects. However, the underlying mechanism by which Avicularin exerts its anti-inflammatory activity has not been clearly demonstrated. This study aimed to elucidate the anti-inflammatory mechanism of Avicularin in lipopolysaccharide (LPS)-stimulated RAW 264.7 macrophage cells.
METHODS AND RESULTS:
Avicularin significantly inhibited LPS-induced excessive production of pro-inflammatory mediators such as nitric oxide (NO) and PGE2 and the protein levels of iNOS and COX-2, which are responsible for the production of NO and PGE2, respectively. Avicularin also suppressed LPS-induced overproduction of pro-inflammatory cytokine IL-1β. Furthermore, Avicularin significantly suppressed LPS-induced degradation of IκB, which retains NF-κB in the cytoplasm, consequently inhibiting the transcription of pro-inflammatory genes by NF-κB in the nucleus. To understand the underlying signaling mechanism of anti-inflammatory activity of Avicularin, involvement of multiple kinases was examined. Avicularin significantly attenuated LPS-induced activation of ERK signaling pathway in a concentration-dependent manner.
CONCLUSIONS:
Taken together, the present study clearly demonstrates that Avicularin exhibits anti-inflammatory activity through the suppression of ERK signaling pathway in LPS-stimulated RAW 264.7 macrophage cells.
Planta Med. 2015 Mar;81(5):373-81.
Pharmacokinetic evaluation of avicularin using a model-based development approach.
The aim of this study was to use the pharmacokinetic information of Avicularin in rats to project a dose for humans using allometric scaling.
METHODS AND RESULTS:
A highly sensitive and specific bioanalytical assay to determine Avicularin concentrations in the plasma was developed and validated for UPLC-MS/MS. The plasma protein binding of Avicularin in rat plasma determined by the ultrafiltration method was 64%. The pharmacokinetics of Avicularin in nine rats was studied following an intravenous bolus administration of 1 mg/kg and was found to be best described by a two-compartment model using a nonlinear mixed effects modeling approach. The pharmacokinetic parameters were allometrically scaled by body weight and centered to the median rat weight of 0.23 kg, with the power coefficient fixed at 0.75 for clearance and 1 for volume parameters. Avicularin was rapidly eliminated from the systemic circulation within 1 h post-dose, and the Avicularin pharmacokinetic was linear up to 5 mg/kg based on exposure comparison to literature data for a 5-mg/kg single dose in rats.
CONCLUSIONS:
Using allometric scaling and Monte Carlo simulation approaches, the rat doses of 1 and 5 mg/kg correspond to the human equivalent doses of 30 and 150 mg, respectively, to achieve comparable plasma Avicularin concentrations in humans.
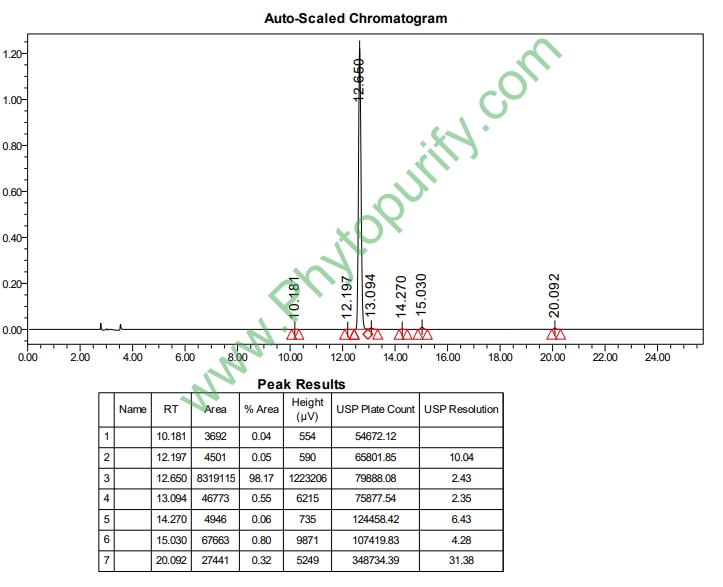
HPLC of Avicularin