
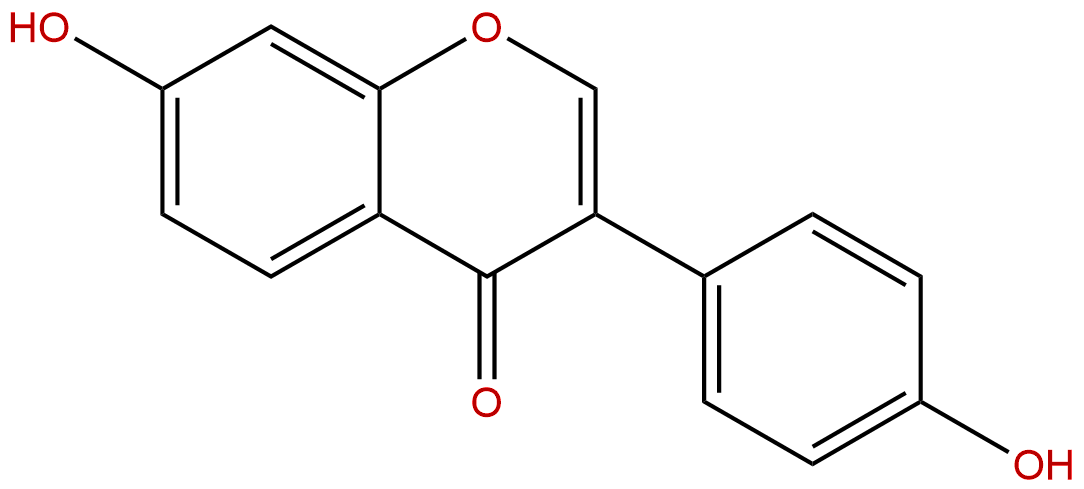
DaidzeinCAS No.:486-66-8
|
||||||||||
 |
|
|
||||||||

| Catalogue No.: | BP0445 |
| Formula: | C15H10O4 |
| Mol Weight: | 254.241 |
Product name: Daidzein
Synonym name:
Catalogue No.: BP0445
Cas No.: 486-66-8
Formula: C15H10O4
Mol Weight: 254.241
Botanical Source: Widespread isoflavone in the Leguminosae (Papilionoideae) e.g. in Chamaecytisus spp., Cytisus spp., Phaseolus spp. Also from Streptomyces xanthophaeus and Streptoverticillium album
Physical Description: Yellow powder
Type of Compound: Flavonoids
Purity: 95%~99%
Analysis Method: HPLC-DAD or/and HPLC-ELSD
Identification Method: Mass, NMR
Packing: Brown vial or HDPE plastic bottle
Storage: Store in a well closed container, protected from air and light. Put into refrigerate or freeze for long term storage.
Whenever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20℃. Generally, these will be useable for up to two weeks.
The product could be supplied from milligrams to grams, up to kilograms
Inquire for bulk scale.
Descriptions:
Daidzein and genistein glucuronides (DG and GG), major isoflavone metabolites, may be partly responsible for biological effects of isoflavones, such as estrogen receptor binding and natural killer cell (NK) activation or inhibition; DG and GG are weakly estrogenic, and they activate human NK cells in nutritionally relevant concentrations in vitro, probably at a site different from IL-2 action.[1]
Daidzein, genistein, and their .beta.-glycoside conjugates can inhibit mammary tumorigenesis in animal models of breast cancer, may have antitumor activity against breast cancer.[2]
Daidzein and daidzin can suppress free-choice ethanol intake by Syrian golden hamsters.[3]
Daidzein or 17 alpha-ethinylestradiol is more efficient than genistein in preventing ovariectomy-induced bone loss in rats.[4]
References:
[1] Zhang Y, Song T T, Cunnick J E, et al. J Nut, 1999, 129(2):399-405.
[2] Coward L, Barnes N C, Setchell K D R, et al. J Agr Food Chem, 1993, 41(11):1961-7.
[3] Keung W M, Vallee B L. P Nat Acad Sci USA, 1993, 90(21):10008-12.
[4] Picherit C, Coxam V, Bennetau-Pelissero C, et al. J Nut, 2000, 130(7):1675-81.
[5] Zhao X, Shen Q, Ma Y. J Chromatogr B, 2011, 879(1):113-6.
HPLC of Daidzein
